Lexaria Bioscience Corp. (CSE:LXX, OTCQX:LXRP), (the “Company” or “Lexaria”) an innovator in drug delivery platforms, reports on its existing technology licensees that have signed definitive contracts to use Lexaria’s revolutionary DehydraTECHTM absorption technology within their existing and emerging brands.
Lexaria Bioscience Corp. (CSE:LXX, OTCQX:LXRP), (the “Company” or “Lexaria”) an innovator in drug delivery platforms, reports on its existing technology licensees that have signed definitive contracts to use Lexaria’s revolutionary DehydraTECHTM absorption technology within their existing and emerging brands.
“The first half of 2019 delivered a period of unprecedented achievements for Lexaria,” said Chris Bunka, Chief Executive Officer. “We signed more license agreements than ever before in our Company’s history, many of which were larger in scope than anything previous. We also launched our brand-new business division Lexaria Nicotine which is disrupting nicotine delivery methods that already attracted one of the world’s largest nicotine companies, which has licensed our technology.”
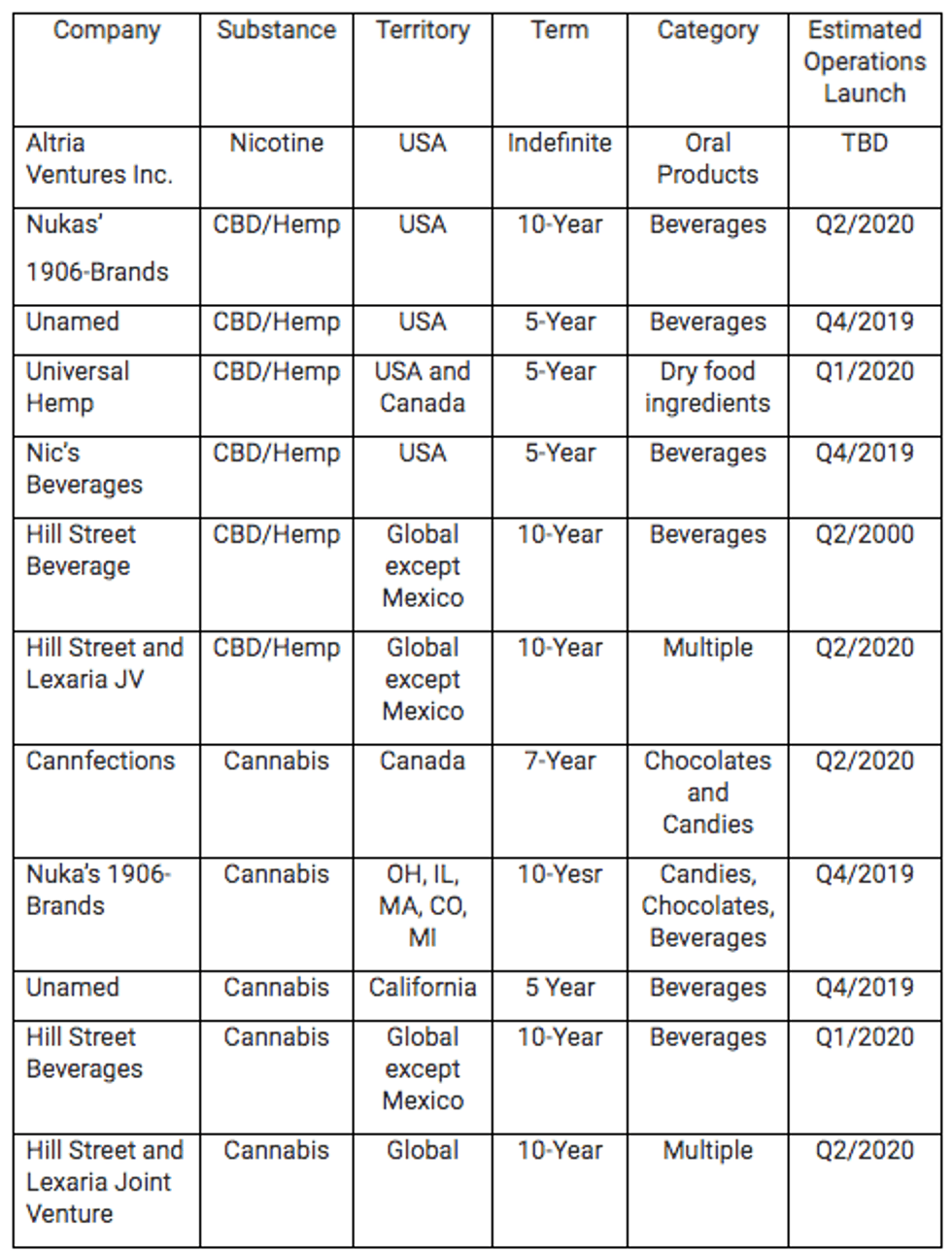
Year to date 2019, Lexaria has announced:
- 1 new license agreement with *Altria Ventures Inc, an indirect wholly-owned subsidiary of Altria Group Inc. for oral nicotine products (note 1);
- 1 new CBD/hemp license to Nuka for beverages across the US;
- 2 new world-wide beverage licenses to Hill Street Beverages (except Mexico);
- 2 new Joint Manufacturing Production licenses with Hill Street Beverages for new jointly owned cannabis and CBD brands;
- 3 new licenses with others for CBD beverages and edible CBD ingredient/products across America.
- 1 new license for cannabis beverages in California;
- 1 new multi-state cannabis expansion license with Nuka Enterprises LLC (“Nuka”) for its 1906 Brands for 5 states total, and 2 new product categories
Lexaria now has 9 corporate licensees who are developing innovative beverages, edibles and oral products with their 11 licenses using the Company’s fully patented DehydraTECH absorption technology:
Based on these existing contracts, Lexaria expects to experience unprecedented revenue growth as we estimate many of our clients will commence operations in late 2019 and early 2020. Several of these clients are already planning and preparing for aggressive roll-outs and growth of their product lines. (Note that estimated operations start dates are Lexaria’s own estimates and may or may not reflect actual licensee results.)
Lexaria credits its recent commercial success mainly to its ever-growing body of scientific results evidencing the superior capabilities and power of its patented technologies. Recent 2019 technological advancements are the result of one of the industry’s most focused and productive R&D programs and hold promise for even higher levels of performance from new DehydraTECH innovations with even faster drug delivery into blood plasma and across the blood-brain-barrier.
In one recent animal study, as earlier announced, Lexaria’s latest patent-pending formulations delivered more than 1,900% more CBD into brain tissue than generic industry formulations and achieved peak blood levels more than 800% greater than those of concentration matched controls. In that same animal study, detection of CBD in the bloodstream began in as little as 2 minutes after dosing and peaked in as little as 45 minutes after dosing providing additional support that DehydraTECH enables rapid onset and offset in line with consumer preferences.
Lexaria expects to sign additional new definitive license agreements through the balance of 2019 and beyond as demand for its industry-leading technology continues, and it commends its family of existing licensees and partners that have already joined the Lexaria ecosystem with their exciting products and offerings powered by DehydraTECH.
*Note 1: The license with Altria Ventures Inc is with Lexaria Nicotine LLC, a separate business division of Lexaria Bioscience Corp.
About Lexaria
Lexaria Bioscience Corp. has developed and out-licenses its disruptive delivery technology that promotes healthier ingestion methods, lower overall dosing and higher effectiveness of lipophilic active molecules. Lexaria has multiple patents pending in over 40 countries around the world and has patents granted in the USA and in Australia for utilization of its DehydraTECHTM delivery technology. Lexaria’s technology provides increases in intestinal absorption rates; more rapid delivery to the bloodstream; and important taste-masking benefits, for orally administered bioactive molecules including cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs), nicotine and other molecules.
FOR FURTHER INFORMATION PLEASE CONTACT:
Lexaria Bioscience Corp.
Alex Blanchard, Communications Manager
(250) 765-6424 ext. 202
Or
NetworkNewsWire (NNW)
www.NetworkNewsWire.com
FORWARD-LOOKING STATEMENTS
This release includes forward-looking statements. Statements which are not historical facts are forward-looking statements. The Company makes forward-looking public statements concerning its expected future financial position, results of operations, cash flows, financing plans, business strategy, products and services, competitive positions, growth opportunities, plans and objectives of management for future operations, including statements that include words such as “anticipate,” “if,” “believe,” “plan,” “estimate,” “expect,” “intend,” “may,” “could,” “should,” “will,” and other similar expressions are forward-looking statements, including but not limited to: that any additional patent protection will be realized or that patent achievements will deliver material results. Such forward-looking statements are estimates reflecting the Company’s best judgment based upon current information and involve a number of risks and uncertainties, and there can be no assurance that other factors will not affect the accuracy of such forward-looking statements. Factors which could cause actual results to differ materially from those estimated by the Company include, but are not limited to, government regulation and regulatory approvals, managing and maintaining growth, the effect of adverse publicity, litigation, competition, scientific discovery, the patent application and approval process and other factors which may be identified from time to time in the Company’s public announcements and filings. There is no assurance that existing capital is sufficient for the Company’s needs or that it will be able to raise additional capital. There is no assurance the Company will be capable of developing, marketing, licensing, or selling edible products containing cannabinoids, nicotine or any other active ingredient. There is no assurance that any planned corporate activity, scientific research or study, business venture, letter of intent, technology licensing pursuit, patent application or allowance, consumer study, or any initiative will be pursued, or if pursued, will be successful. There is no assurance that any of Lexaria’s postulated uses, benefits, or advantages for the patented and patent-pending technology will in fact be realized in any manner or in any part. No statement herein has been evaluated by the Food and Drug Administration (FDA). Lexaria-associated products are not intended to diagnose, treat, cure or prevent any disease.
Source: www.accesswire.com
The Conversation (0)
Latest News
Outlook Reports world
Featured Cannabis Investing News Stocks
Browse Companies
MARKETS
COMMODITIES
CURRENCIES