RAMM Pharma (CSE:RAMM) begins trading on the Canadian Securities Exchange today under the ticker symbol “RAMM”.
RAMM Pharma Corp. (including its wholly owned subsidiaries, the “Company” or “RAMM”) (CSE:RAMM), a leader in plant-derived cannabinoid pharmaceutical products, is pleased to announce that trading in its common shares on the Canadian Securities Exchange will commence today at 9:30 a.m. (EST) under the ticker symbol “RAMM”.
RAMM is one of the leaders in the field of cannabinoid pharmacology and product formulation for cannabis-derived prescription drugs and registered products. Founded in 1988 in Montevideo, Uruguay, RAMM is an established pharmaceutical and medical product business. In 2017 the Company received approval for its first plant-derived cannabinoid pharmaceutical drug from Uruguay’s Ministry of Health and now has a variety of approved and registered products that have been authorized for sale in Uruguay and on a compassionate use basis in several other Latin American countries, as well as a pipeline of new products in various stages of approval and development. RAMM’s formulation and manufacturing is conducted at its state-of-the-art Good Manufacturing Practices (GMP) certified cannabis formulation facility ideally situated within close proximity to an international airport and other export hubs. The facility totals approximately 36,600 sq.ft. and features dedicated cannabis and medical product laboratories along with packaging capabilities and storage/distribution facilities
The Company is also in the process of establishing vertically integrated operations through the development of its large-scale cultivation facility. In addition to RAMM’s industry leading activities in the field of cannabinoid pharmaceuticals, RAMM also operates a successful pharmaceutical, cosmetic and nutraceutical product development and medical services business which has been in operations for 30 years.
“Our public listing marks a significant milestone as RAMM continues to establish itself as a leader in the development and commercialization of cannabis-derived prescription drugs and registered products to meet the growing demand in Latin America and other jurisdictions globally,” stated Jack Burnett, Chief Executive Officer. “We are very pleased with the interest in our suite of products and look forward to increasing access to their therapeutic benefits as we continue to expand our product offerings, awareness and availability.”
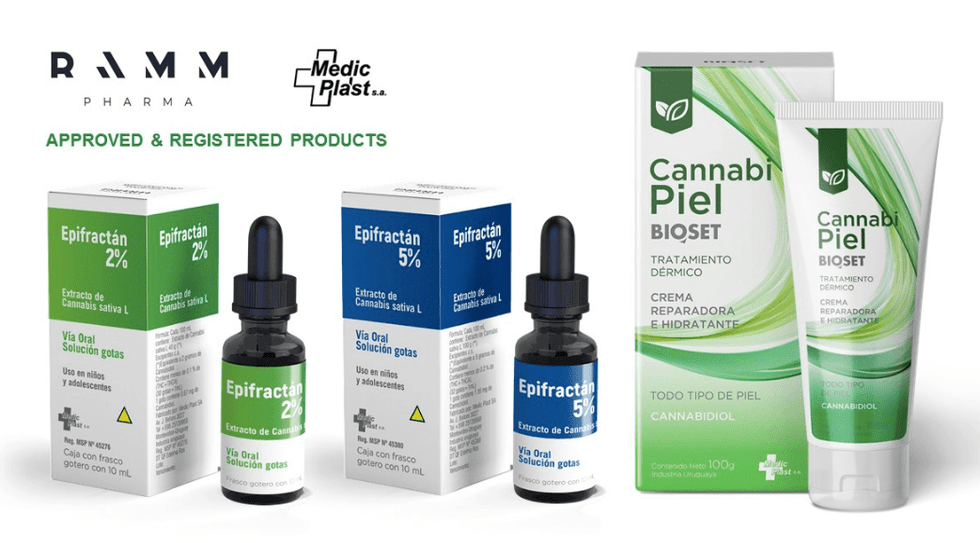
RAMM currently sells five cannabis based registered products commercially in Uruguay under two brands – Epifractán™ and CannabiPiel™. RAMM’s products have also been approved for use on a compassionate basis in 3 other Latin American countries and are in the application process at various stages of approval in several other countries. Uruguay’s stable economy, currency, political climate and strong pharmaceutical research sector make it an ideal base for product research and development and to service the Latin American population of approximately 640 million people.
About Epifractán™
In 2017, Epifractán™ became the first plant-derived cannabinoid pharmaceutical drug to be approved by a Latin American federal health authority. The drug is currently sold commercially in 2% and 5% cannabidiol (CBD) concentrations. A 10% CBD concentration is in the process of being approved by the Ministry of Health (MSP). Epifractán™ is an approved treatment for refractory epilepsy with a clinical trial about to commence examining its efficacy for the treatment of chronic pain.
About CannabiPiel™
CannabiPiel™ is a cosmeceutical cream with active ingredient 1% CBD. It is available over the counter and is used for the treatment of joint pain and inflammation, as well as a variety of skin disorders and conditions. Its unique Nioskin structure makes for optimal delivery of CBD properties.
Medical Supplies/Device Business
RAMM has an existing medical supplies and device business that currently generates sufficient revenue to cover the burn rate associated with this segment of RAMM’s business. Furthermore, these operations provide access and have developed critical relationships within the medical professional, regulator and hospital communities in Uruguay.
Subscription Receipt Financing
In connection with its public listing RAMM has recently completed a non-brokered private placement for aggregate gross proceeds of approximately C$35.3 million at a price of $1.35 per subscription receipt. Upon the completion of the listing transactions, the subscription receipts were converted on a one-for-one basis into a total of 26,165,109 RAMM Shares on October 28, 2019.
Directors, Officers & Management
Jack Burnett (Chairman, Chief Executive Officer and Director)
Mr. Burnett is a successful entrepreneur with over 40 years’ experience in the capital markets and international corporate leadership roles. Mr. Burnett has led companies from inception to acquisition in multiple industries including real estate, insurance and telecom. His deep global business relationships span both private and public markets where he has been a director, officer and majority shareholder of successful multinational companies.
Guillermo Delmonte (Chief Operating Officer)
Mr. Delmonte is a Certified Public Accountant from the ORT University of Uruguay and holds a Financial Advisor certificate from the Institut d’Estudis Financers Barcelona, Spain. Mr. Delmonte has over five years of experience in management and business development of cannabis in the international markets. Prior to joining RAMM, Mr. Delmonte was the President of the International Division of Organigram Inc. (TSX.V: OGI), a major Canadian licensed producer of cannabis, where he successfully oversaw Organigram’s first international expansion. Prior to its acquisition by Aurora Cannabis Inc. (NYSE: ACB), Mr. Delmonte served as Director and Chief Executive Officer at ICC Labs, the first South American cannabis company to go public. Mr. Delmonte has also held financial and business development leadership roles at companies including BBVA Bank, Technit Group, Credit Agricole Bank, United Nations Uruguay and Bank ITAU.
Matias Piñeiro (Chief Financial Officer)
Mr. Piñeiro is a public accountant and experienced finance professional with a demonstrated history of directing comprehensive finance and accounting operations and providing leadership. Skilled in technology, financial reporting, budgeting and financial statements auditing, with a focus on driving operational performance, profitability, controls and continuous improvement. From 2017 to 2019, Mr. Piñeiro served as the accounting and reporting line manager for Syngenta AG, a global company that produces agrochemicals and seeds and is based in Basel, Switzerland, from 2016 to 2017 served as corporate financial controller for a subsidiary of UG International Holdings operating in the agriculture, renewable energy, real estate and cannabis markets in South America. From 2012 to 2016, Mr. Piñeiro was an audit manager with KPMG, and is currently an assistant professor of FS Audit and Professional Reports of Public Accountants at the Faculty of Economics Sciences and Administration at Universidad de la República Oriental del Uruguay (UDELAR).
Dr. Armando Blankleider (Director)
Dr. Blankleider is a Medical Doctor and the founder and President of Medic Plast. Dr. Blankleider has directly led Medic Plast’s initiatives for the design and introduction of new products, as well as in the design and monitoring of teams for the development of production processes and the general management of Medic Plast. Dr. Blankleider also has a depth of experience in Quality Management ISO Standards, has acted as a delegate to develop the Rules of Good Manufacturing Practices for medical products for the private sector within the MERCOSUR and is an active participant in international conferences for the medical and pharmaceutical products industry globally.
Dr. Leticia Cuñetti (Chief Medical Officer)
Dr. Cuñetti is a Medical Doctor, nephrologist and pharmacologist. She holds a Masters degree in Donation and transplantation of organs and tissues from the Universidad de Barcelona, Spain, and is a fellow of the International Society of Nephrology. She served as nephrology assistant from 2006 to 2012 and as Associate professor of Pharmacology in the Medical School of the Universidad de la Republica, Uruguay, from 2000 to 2017. Currently Dr. Cuñetti is the Vice-President of the Uruguayan Endocannabinoid Society (SUEN).
Edelma Ros (Chief Technical Director)
Ms. Ros is a Pharmaceutical Chemist specializing in Pharmaceutical Care at the Universidad de la Republica, Uruguay. Ms. Ros is a proven specialist in the pharmaceutical and cosmetic formulation industry, leading Medic Plast’s technical direction for more than 15 years and has created more than a hundred registered pharmaceutical products.
Daniel Augereau (Director)
Mr. Augereau is a seasoned executive who has held senior leadership and board-level positions at companies spanning a diverse mix of industries over a 50+ year career. Since 2005, Mr. Augereau has served as the Chairman and Chief Executive Officer of Synergie SA (Euronext: SDG), the French leader of temporary work and human resources management services for the industry, tertiary, logistics, medical, building and public works sectors.
Eric Klein (Director)
Mr. Klein focuses on complex mergers, acquisitions, divestitures, financings as well as joint ventures and business valuations of mid-sized Canadian corporations. With more than 30 years of experience, he focuses on providing results-driven corporate finance services for mid-market Canadian companies. Recently, Mr. Klein was a senior executive with Dundee Corporation. Prior to that Mr. Klein was the founder and Managing Director of the Corporate Finance, Valuations & Transaction practice of Farber Financial Group, a consulting company. Mr. Klein is a graduate of McGill University with a B.Comm and a graduate Diploma in Public accounting, and holds designations as a Chartered Public Accountant and Chartered Business Valuator.
Matthew Bajurny (Director)
Mr. Bajurny is a CPA with a Bachelor of Commerce in Accounting from the University of Guelph and brings to the board strong financial literacy skills further developed through over his years of professional experience in financial statement audit at public accounting firms PwC LLP and Crowe Mackay LLP and as the CFO of a publicly listed Canadian corporation and other directorship positions.
About RAMM Pharma Corp.
Lead by renowned cannabis industry experts and backed by successful pioneers in the cannabis sector, RAMM is a leader in the field of cannabinoid pharmacology and product formulation for cannabis-based pharmaceuticals and other cannabis-based products. Founded in 1988 in Montevideo, Uruguay, the Company is a well established pharmaceutical and medical product business that has developed medically registered and approved plant-derived cannabinoid pharmaceutical products. The Company currently has multiple approved and registered products that have been authorized for sale in Uruguay and compassionate use in several Latin American countries, as well as a pipeline of new products in various stages of approval and development produced in the Company’s state of the art Good Manufacturing Practice (GMP) certified cannabis formulation facility. With its large-scale cultivation facility, the combined operations are expected to provide for complete vertical integration. Further to its industry leading activities in the cannabis sector, the Company operates a successful pharmaceutical, cosmetic and nutraceutical product development and medical services business which has been servicing the local market for 30 years.
RAMM Pharma Corp. includes wholly owned subsidiaries Medic Plast SA, Yurelan SA and Ramm Pharma Holdings Corp.
Additional information about the Company is available at www.rammpharma.com.
For further information, please contact:
Guillermo Delmonte
Chief Operating Officer
(484)-474-0932
info@rammpharma.com
Cautionary Note Regarding Forward-Looking Information
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward looking statements”) within the meaning of the applicable Canadian securities legislation. All statements, other than statements of historical fact, are forward-looking statements and are based on expectations, estimates and projections as at the date of this news release. Any statement that involves discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, assumptions, future events or performance (often but not always using phrases such as “expects”, or “does not expect”, “is expected”, “anticipates” or “does not anticipate”, “plans”, “budget”, “scheduled”, “forecasts”, “estimates”, “believes” or “intends” or variations of such words and phrases or stating that certain actions, events or results “may” or “could”, “would”, “might” or “will” be taken to occur or be achieved) are not statements of historical fact and may be forward-looking statements. In this news release, forward looking statements relate, among other things, the Company’s strategies and objectives, and future expansion plans.
These forward-looking statements are based on reasonable assumptions and estimates of management of the Company at the time such statements were made. Actual future results may differ materially as forward-looking statements involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements of the Company to materially differ from any future results, performance or achievements expressed or implied by such forward-looking statements. Such factors, among other things, include: the commencement of trading of the common shares of RAMM on the Canadian Securities Exchange; fluctuations in general macroeconomic conditions; fluctuations in securities markets; expectations regarding the size of the Uruguayan, Latin American, and international medical and recreational cannabis markets and changing consumer habits; the ability of the Company to successfully achieve its business objectives; plans for expansion; political and social uncertainties; inability to obtain adequate insurance to cover risks and hazards; and the presence of laws and regulations that may impose restrictions on cultivation, production, distribution and sale of cannabis and cannabis related products in Uruguay or internationally; and employee relations. Although the forward-looking statements contained in this news release are based upon what management of the Company believes, or believed at the time, to be reasonable assumptions, the Company cannot assure shareholders that actual results will be consistent with such forward-looking statements, as there may be other factors that cause results not to be as anticipated, estimated or intended. Readers should not place undue reliance on the forward-looking statements and information contained in this news release. The Company assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change, except as required by law.
No stock exchange, securities commission or other regulatory authority has approved or disapproved the information contained herein.
The Conversation (0)
Latest News
Outlook Reports world
Featured Cannabis Investing News Stocks
Browse Companies
MARKETS
COMMODITIES
CURRENCIES