Australian Cannabis Players Partner Up with Supply Agreement
MGC Pharma will be tasked with providing THC Global with medical cannabis products to be branded under THC Global’s wholly owned subsidiary.
Medical cannabis companies THC Global Group (ASX:THC) and MGC Pharmaceuticals (ASX:MXC,OTCQB:MGCLF) are teaming up through a new supply agreement.
Monday’s (January 20) announcement confirms that MGC Pharma will be tasked with providing THC Global with additional medical cannabis products, to be branded under the Canndeo name, which is THC Global’s wholly owned subsidiary.
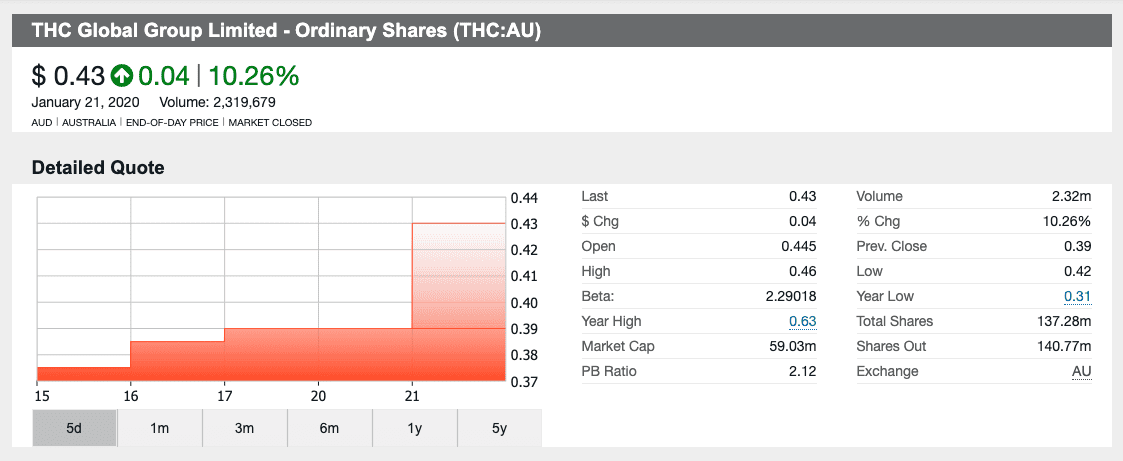
THC Global shares surged across Monday’s trading halt as the deal closed. The company was up over 14 percent at market open on Tuesday (January 21) from the end of the trading session on Friday (January 17).
With this new deal in place, THC Global plans to distribute products across Australia and New Zealand during Q1 2020. The company has already made its first purchase order, valued at 25,000 euros, with future orders worth at least 50,000 euros.
For the initial product launch, THC Global will offer three oral liquid formulations with varying levels of tetrahydrocannabinol (THC) and cannabidiol (CBD). The deal, signed with an initial term of 18 months, also gives the two companies space to expand the contract to include cannabis extracts and cannabis-based active pharmaceutical ingredients.
In a press release, Ken Charteris, CEO at THC Global, said the deal allows the firm to leverage its production capacity at its facility in Southport, Queensland — the largest pharmaceutical extraction facility in the Southern Hemisphere, according to THC Global — to expand its activities across Australia, New Zealand, Canada and Asia.
The agreement also gives THC Global the chance to pivot to using Australia-produced cannabis and away from the international imports that have powered the medical marijuana industry in Australia thus far as companies in the country finally bring in the yields from their first harvests.
Roby Zomer, CEO of MGC Pharma, said in the release that the deal, and future agreements between the two firms, will serve to boost MGC Pharma’s international presence as it develops its production and research facility in Malta.
The supply deal comes shortly after the firm announced its first “farm-to-pharma” processing of medical cannabis in December at its facility in Southport. The deal is also a part of THC Global’s recent push to expand its reach across Australia and international markets.
On Tuesday (January 21), the Southport facility was granted a good manufacturing practices licence to manufacture therapeutic goods, defined as products that prevent, cure or alleviate an illness, among other effects, from the Therapeutic Goods Administration.
With the certification, THC Global said it’s now fully licenced to create medical cannabis products in Australia, giving it the ability to produce cannabis extracts, package medical marijuana products and supply medicinal cannabis for human trials in Australia, as well as globally in countries across Europe, Asia and North America.
Don’t forget to follow us @INN_Australia for real-time news updates!
Securities Disclosure: I, Danielle Edwards, hold no direct investment interest in any company mentioned in this article.