- WORLD EDITIONAustraliaNorth AmericaWorld
Investor Insight
Cardiex is trailblazing the health technology industry through its innovative solutions that are transforming cardiovascular health care.
Overview
Cardiex Limited (ASX:CDX) is an ASX-listed medical technology company leveraging its proprietary SphygmoCor® technology to develop and market vascular biomarker technologies and digital solutions focused on the world’s largest health disorders. The company’s groundbreaking technology – SphygmoCor® – set the benchmark for noninvasive measurement of central aortic pressures and related arterial health characteristics, collectively referred to as vascular biomarkers.
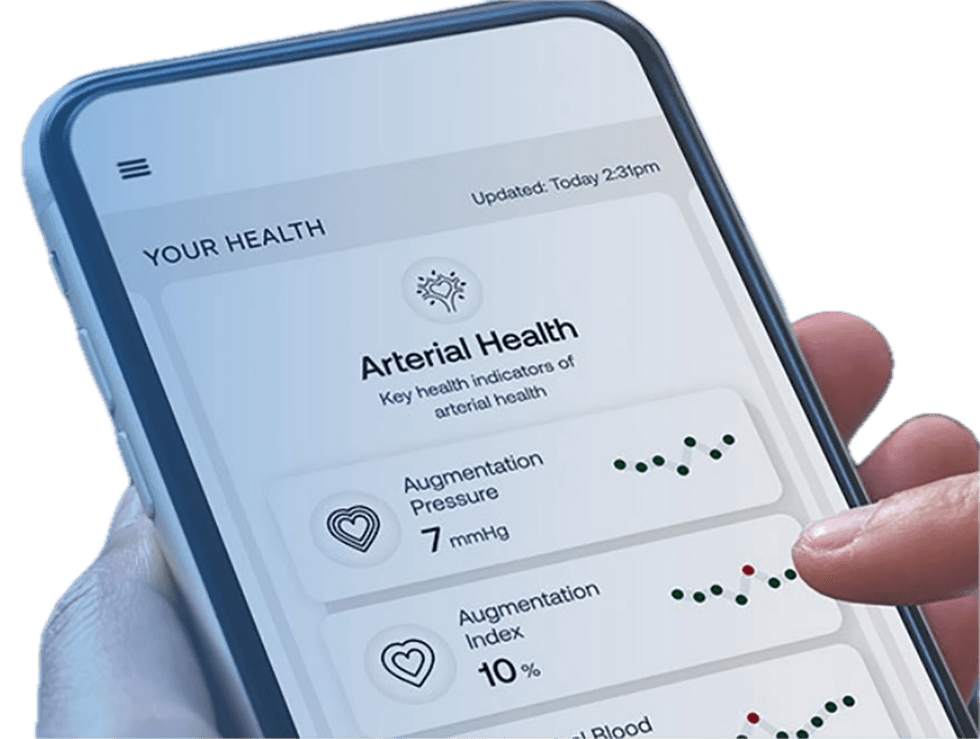
While measuring arterial health parameters has always been considered clinically beneficial, it was not considered for routine out-patient clinical use as it previously required an invasive catheterization procedure with a pressure sensor inserted into the aorta. Founded on 40 years of hemodynamics studies and backed by 20+ years of research, Cardiex’s SphygmoCor technology employs non-invasive techniques to assess "central aortic waveforms," offering valuable insights into various clinically significant arterial health parameters including arterial stiffness, central blood pressure (the pressure at the heart), pulse pressure, and crucial indicators of vascular health for major organs such as the heart, brain and kidneys.
Assessing central blood pressure directly at the heart is deemed superior to conventional blood pressure measurements taken at the arm, primarily owing to the heart's proximity to vital organs. Cardiex’s FDA-cleared devices replace traditional blood pressure technology for first-line screening and monitoring of arterial health status. The unique physiologic insights from the company’s devices provide clinically relevant information that helps guide treatment decisions and offers profound benefits for all members of the healthcare community:
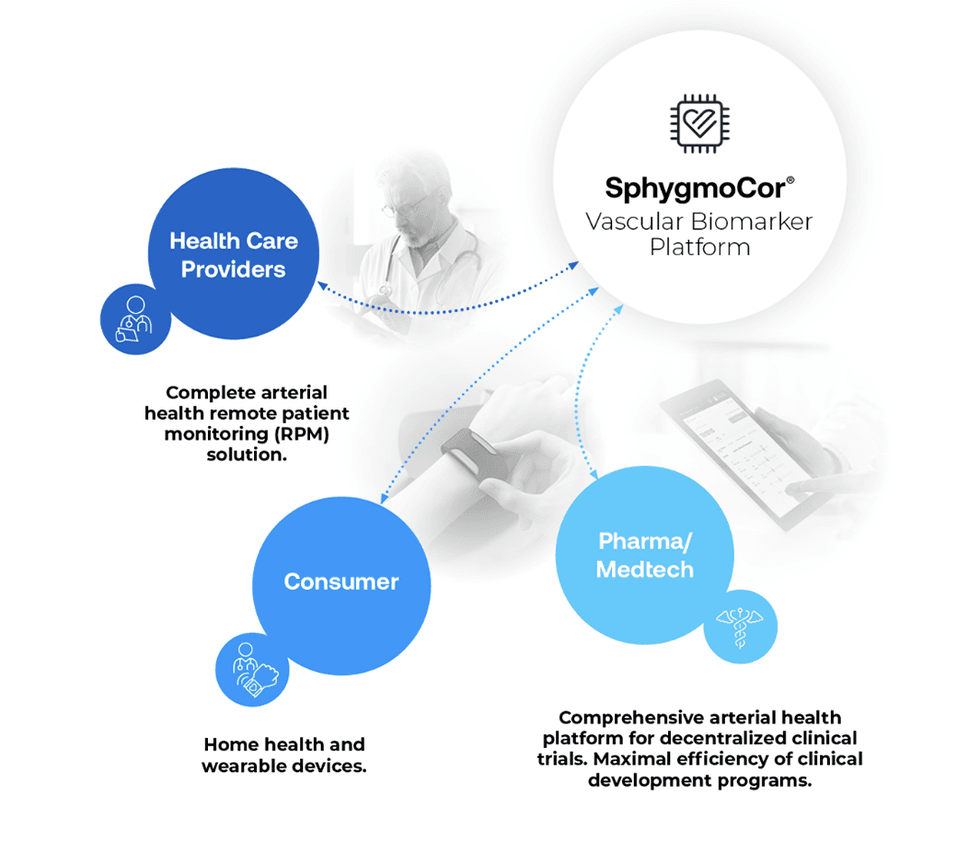
For Healthcare Providers: Enable physicians to make more informed treatment decisions based on clinically relevant vascular health data.
For Patients: Give patients the tools to make better decisions about their own health.
For Pharmaceutical Companies: Generate reliable, real-world, clinically relevant data to accelerate drug development and commercialization.
For more than two decades, the company’s SphygmoCor technology has set the benchmark for vascular biomarker assessments, adopted by premier hospitals and pharmaceutical giants worldwide. SphygmoCor is the chosen technology for measuring central blood pressure in all of the "top 20 hospitals" in the US and has played a crucial role in the clinical trials of leading firms including Bayer, AstraZeneca, Roche, Novartis and GlaxoSmithKline.

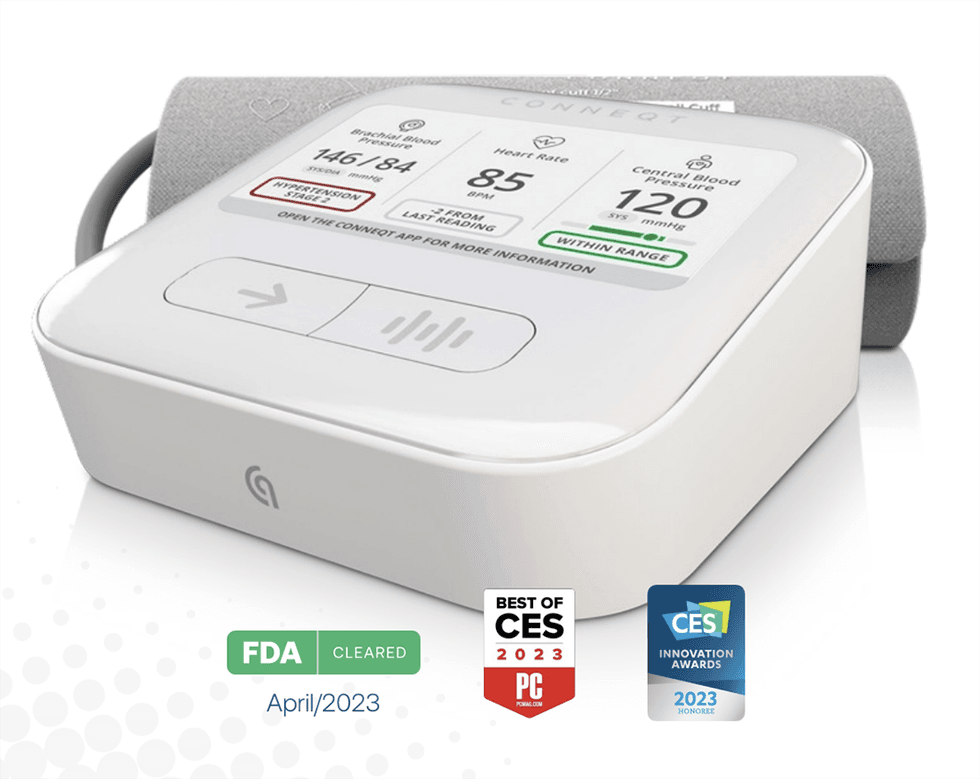
Between 2002 and 2023, CDX received five FDA clearances, the latest for the CONNEQT Pulse, a first-of-its-kind connected vascular biometrics monitor.
Cardiex devices have previously been sold and used exclusively in clinical settings – principally by specialist clinicians, for research, and by pharmaceutical companies for drug assessment.
The introduction of CONNEQT Pulse represents a significant shift for Cardiex, allowing the company to enter the connected care market and transition from niche segments to the mass healthcare market. Priced comparably to a home health monitor, the CONNEQT Pulse is positioned for widespread adoption and can be deployed at scale in general healthcare practices, homes, or any location where patients are present.
The CONNEQT Pulse will also bolster Cardiex's portfolio in clinical trial solutions with the introduction of a decentralized clinical trial (DCT) platform. The Cardiex DCT platform empowers clinical trial managers to remotely monitor thousands of patients in their homes, enabling pharmaceutical companies to more effectively evaluate potential vascular outcomes across various trial phases. This leads to increased efficiency and cost-effectiveness in trial outcomes.
Furthermore, in response to the growing demand for proactive health monitoring, Cardiex will be launching an innovative wrist-worn device that leverages the SphygmoCor technology to deliver a medical grade wearable with capabilities far beyond conventional health trackers. The CONNEQT Band will be a wearable “cuffless” device designed to monitor vascular health in patients as well as to provide general health insights to consumers.
Cardiex’s goal is to establish a holistic ecosystem that promotes cardiovascular well-being and empowers users to proactively manage their health as an integral part of individuals' health routines, contributing to a paradigm shift in preventive cardiovascular care.
The company’s first-mover advantage and exclusive technology FDA-cleared for noninvasive measurement of central pulse pressures and vascular biomarkers across all adult demographics grant it a distinctive market position. CDX recently secured AU$14 million in funding, which is enough to steer the company towards profitability.
With the entry into the connected care home market with the CONNEQT Pulse, CDX is transitioning towards a recurring revenue model based on monthly subscription fees. This should excite investors, given that the recurring revenue model will receive a higher multiple by the market, thereby boosting the company's valuation.
Get access to more Medical Device Investing Stock profiles here


