
- NORTH AMERICA EDITIONAustraliaNorth AmericaWorld
December 18, 2024
Founded by a team of seasoned entrepreneurs and healthcare experts, Zero Candida (TSXV:ZCT) is a fem-tech pioneer, combining advanced artificial intelligence with non-drug diagnostics and personalized treatment modalities in a single device. The company developed an innovative, first-of-its-kind solution to the diagnosis and treatment of Candidiasis, a fungal infection causing irritation, discharge and intense itchiness of the vagina and the vulva. The device offers precision therapy by eliminating fungal infections with over 99.99 percent effectiveness in just three hours, a revolutionary improvement over existing treatments.
Zero Candida is on track to complete clinical trials and file for FDA approval in 2024 with its innovative technology.
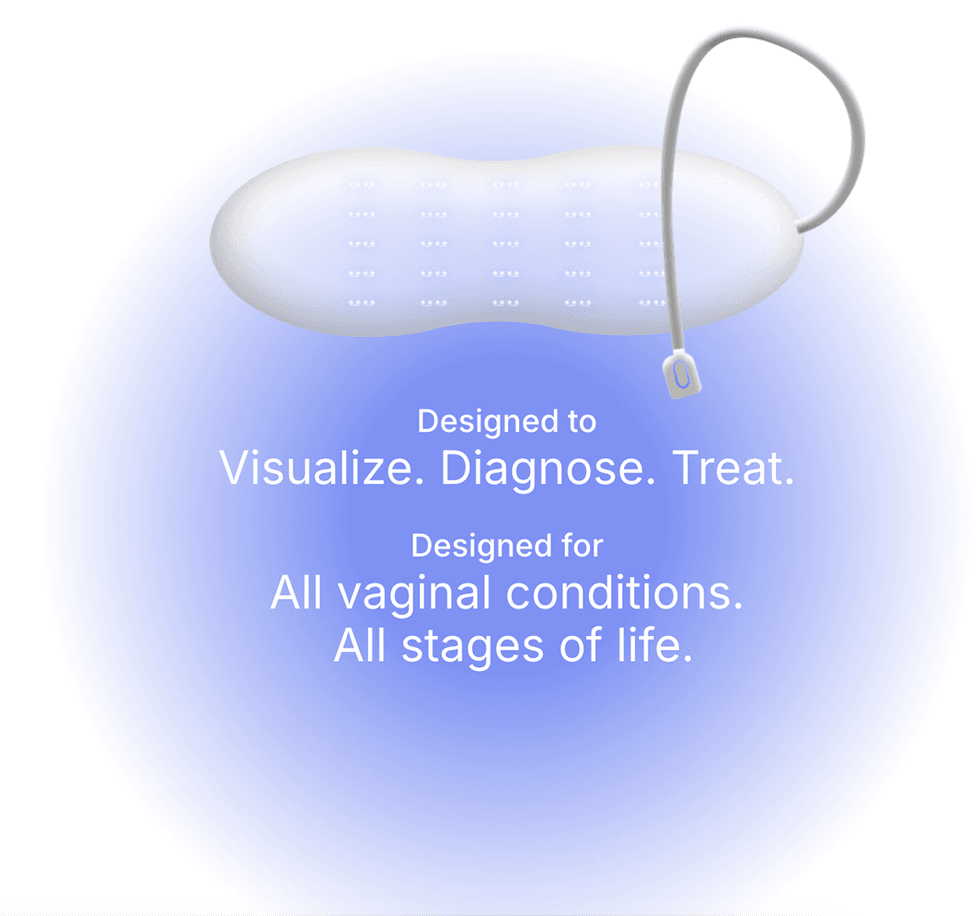
Zero Candida's SMART vaginal diagnostic device utilizes blue light therapy to treat this widespread condition without drugs. This non-drug therapy addresses key issues associated with conventional antifungal medications, including reduced risk of drug resistance, minimized side effects, and potential for faster symptom relief
Company Highlights
- Zero Candida Technologies is a fem-tech company focused on developing a SMART diagnostic and therapeutic device aimed at eliminating vaginal candidiasis (commonly known as yeast infection), a condition that affects three out four women globally.
- Candidiasis a fungal infection causing irritation, discharge and intense itchiness of the vagina and the vulva. In several cases, the current treatment for Candidiasis has been ineffective.
- Zero Candida has completed proof-of-concept studies, and demonstrated near-complete fungal eradication with over 99.99 percent effectiveness in just three hours.
- Founded by a team of experienced entrepreneurs and healthcare experts, the company is addressing the significant unmet needs of the women’s health market.
- The fem-tech segment of the med tech market is expected to grow at a CAGR of 18.2 percent and is estimated to reach nearly US$30 billion by 2032. North America dominated the global fem-tech market with a share of 52.91 percent in 2023.
- The company has already patented this technology in South Africa, while additional filings for patent application in the US and EU are underway.
This Zero Candida profile is part of a paid investor education campaign.*
Click here to connect with Zero Candida (TSXV:CZT) to receive an Investor Presentation
ZCT:CC
The Conversation (0)
16 December 2024
Zero Candida Technologies
Pioneering a technology-driven, innovative solution for the non-drug treatment of Candidiasis.
Pioneering a technology-driven, innovative solution for the non-drug treatment of Candidiasis. Keep Reading...
03 February
Moore Market analysis: Zero Candida potential market will reach over $2 Billion by 2030
According to the report: u p to 75% of women will have at least one vaginal yeast infection in their life - A market analysis made by Moore Financial Consulting published today estimate the global market for Zero Candida Technologies, Inc. (TSXV: ZCT) (FSE: 9L2) (the "Company" or "ZCT"), an... Keep Reading...
25 November 2024
Zero Candida Technologies Inc. Opens the Market
Eli Ben-Haroosh, Co-Founder and Chief Executive Officer, Zero Candida Technologies Inc. (TSXV: ZCT) ("Zero Candida" or the "Company"), joined Dani Lipkin, Managing Director, Global Innovation Sector, Toronto Stock Exchange, to open the market to celebrate the Company's new listing on the TSX... Keep Reading...
18 November 2024
Zero Candida Applies to List its Shares on the TSX Venture Exchange
Zero Candida Technologies Inc. (the "Company" or "Zero Candida") (TSXV: ZCT), a technology company focused on development and commercial exploitation of a number of patents and patent applications with respect to an AI smart tampon-like device, designed to treat Candidiasis and transfer the... Keep Reading...
09 September
How to Invest in Medical Device Stocks and ETFs
The medical device market offers investors unique exposure to the overall life science space, especially in an era of fast-growing tech advancements in healthcare.This industry covers a wide range of health and medical instruments and equipment used in the treatment, mitigation, diagnosis and... Keep Reading...
13 March
BlinkLab Completes First Patient Test for US Autism Diagnostic Study
Digital healthcare company BlinkLab (ASX:BB1) has tested the first patient in its US autism diagnostic study, which is geared at validating the company's Dx1 test as a diagnostic aid for clinicians. BlinkLab states in its Wednesday (March 12) release that the study is the largest digital... Keep Reading...
25 February
HeraMED Signs Strategic Collaboration Agreement with Garmin Health
HeraMED Limited (ASX: HMD), a medical data and technology company leading the digital transformation of maternity care, is delighted to announce it has entered into a collaboration agreement with Garmin (NYSE: GRMN), a leading global provider of smartwatches and GPS-enabled products, aimed at... Keep Reading...
17 February
2 Biggest Medical Device ETFs in 2025
Exchange-traded funds (ETFs) are a popular investment strategy, and generally contain a variety of publicly traded companies under one stock symbol, often with a focus on a specific sector.Depending on the ETF, investors may be able to track up-and-coming companies, get exposure to top firms or... Keep Reading...
23 January
Cyclopharm Signs US Agreement with HCA Healthcare for Technegas®
Cyclopharm Limited (ASX: CYC) is pleased to announce the signing of a major contract with Hospital Corporation of America Healthcare (HCA), one of the largest single healthcare providers in the United States. This agreement marks a significant milestone for the company which will allow the... Keep Reading...
23 January
CONNEQT App Launches in USA as Pulse Deliveries Commence
Cardiex Limited (CDX:AU) has announced CONNEQT App Launches in USA as Pulse Deliveries CommenceDownload the PDF here. Keep Reading...
Latest News
Latest Press Releases
Related News
TOP STOCKS
American Battery4.030.24
Aion Therapeutic0.10-0.01
Cybin Corp2.140.00







