
Awakn Life Sciences Corp.
Revolutionary treatments for addiction
Overview
Awakn Life Sciences (NEO:AWAKN,OTCQB:AWKNF) is a revenue-generating biotechnology company conducting research, development and commercialization of therapeutics to treat addiction. With a near-term focus on alcohol use disorder (AUD), Awakn is the first company in the world to provide evidence-backed ketamine-assisted therapy for AUD. In the medium-term, the company will generate revenue or create value through out-licencing or sale of R&D assets.
Approximately 1 in 8 people in the world are living with a mental disorder, according to data from the World Health Organization. Canada’s Centre for Addiction and Mental Health says people with a mental illness are twice as likely to have a substance use disorder, compared to the general population, and at least 20 percent of those living with a mental illness have a co-occurring substance use disorder.
About 400 million globally are affected by AUD, and yet it remains one of the biggest unmet medical needs today. Awakn is working to disrupt this underperforming industry — currently valued at $25 billion per year — by advancing next-generation drugs and therapies, through preclinical research and clinical stage trials.
The company’s business model focuses on two things: 1) research and development, by combining existing drugs with Awakn’s proprietary therapies to treat AUD; 2) commercialization, through Awakn-owned clinics throughout Europe and through licensing partnerships with third-party addiction treatment clinics in North America.
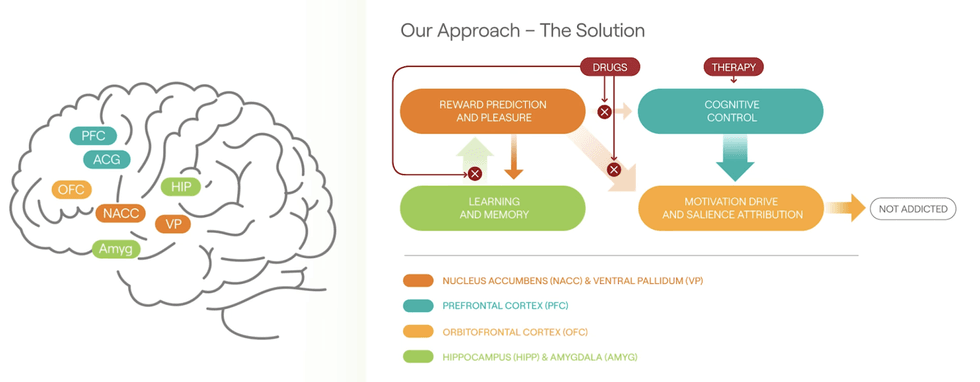
Project Kestrel is the lead clinical development program of Awakn Life Sciences. Project Kestrel is supported by Awakn’s Phase II a/b ‘KARE’ clinical trial which examined ketamine-assisted therapy for the treatment of alcohol use disorder (AUD). The trial resulted in patients experiencing on average 86 percent abstinence at six months post-treatment versus 2 percent before the trial, which means study participants went from being sober on average seven days a year to being sober on average 314 days a year. The company was recently successful in securing funding for Phase III clinical trial for Project Kestrel from the UK’s National Institute for Health and Care Research (NIHR). The NIHR grant will cover 66 percent of the approximately $3.75 million cost of the clinical trial.
Awakn has also successfully completed the world’s first ketamine treatment study for behavioural addictions, led by Professor Celia Morgan, a University of Exeter professor and head of Awakn’s ketamine-assisted therapy. The study yielded promising results for a potential new treatment for behavioural addictions and the company has since initiated a larger study for further investigation. Following the study, Awakn also filed a patent application for exclusive rights to use ketamine and ketamine-assisted therapy for behavioural addictions.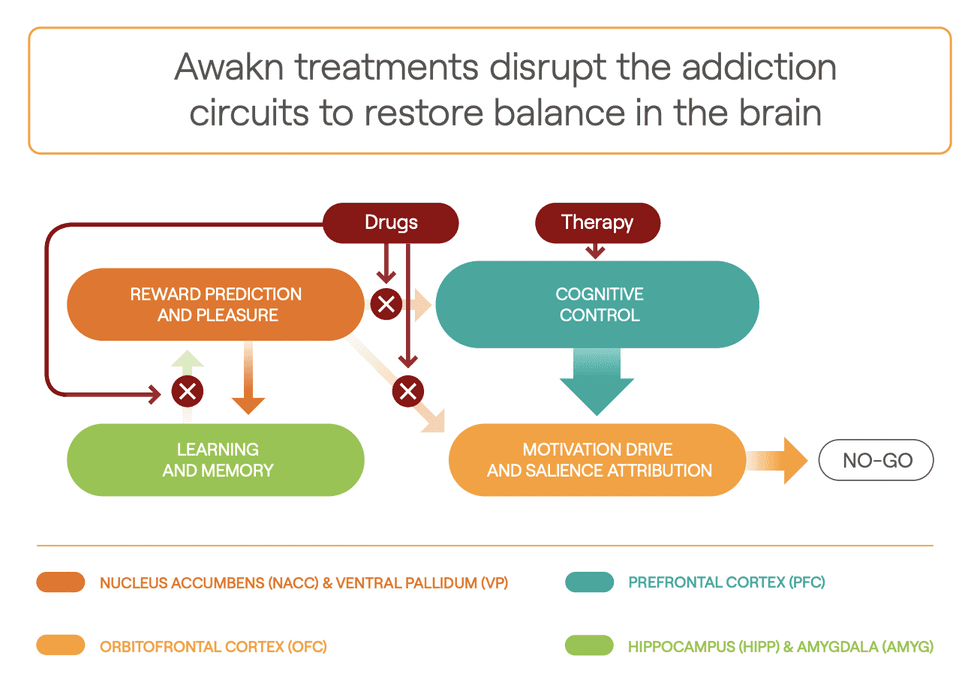
Company Highlights
- Awakn Life Sciences is a revenue-generating biotechnology company researching, developing, and commercializing therapeutics to treat addiction with a near-term focus on Alcohol Use Disorder
- Phase III clinical trial of Awakn’s proprietary ketamine-assisted therapy treatment for AUD has received 66 percent grant funding from the UK state National Institute for Health and Care Research (NIHR)
- Completed world’s first ketamine treatment study for behavioural addictions
- Three operational clinic locations: London and Bristol in the UK, and Oslo in Norway
- Licensing agreements with Revitalist in the US and Wellbeings in Canada for use of Awakn’s proprietary ketamine-assisted therapy for AUD treatment.
- Awakn has a world-leading scientific team with decades of experience researching, developing and commercializing therapeutics to treat addiction
Get access to more exclusive Psychedelics Investing Stock profiles here











