August 13, 2024
TrivarX Limited (‘the Company’) (ASX: TRI) is pleased to announce positive top-line results from the Company’s recently completed Phase 2 Sleep Signal Analysis for Current Major Depressive Episode (SAMDE) study utilising its proprietary AI-backed algorithm, MEB-001. MEB-001 uses EEG and ECG signals recorded during sleep to identify current Major Depressive Episode (cMDE).
Highlights:
- Analysis of 400 patients in Phase 2 SAMDE study for current Major Depressive Episode (cMDE) study utilising MEB-001
- MEB-001 is TrivarX’s proprietary AI-driven algorithm which can assist with the effective screening of a cMDE
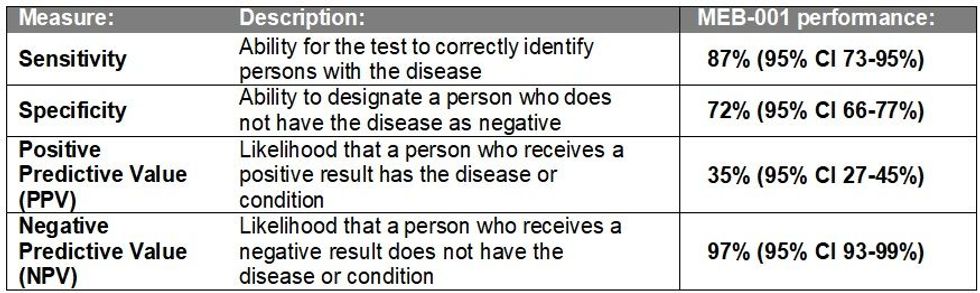
- MEB-001 performance results from Phase 2 SAMDE study reported high performance, marking a significant achievement in the Company:
- Sensitivity: 87% (95% CI 73-95%)
- Specificity: 72% (95% CI 66-77%)
- Positive Predictive Value: 35% (95% CI 27-45%)
- Negative Predictive Value: 97% (95% CI 93-99%)
- Results provide strong validation of MEB-001’s ability to assist in the screening and diagnosis of a cMDE – a condition which is commonly misdiagnosed
- There is currently no screening undertaken for cMDE in sleep centres in the US or globally, representing a major commercial opportunity for TrivarX
- Results advance TRI’s aim to become the first depression screening tool used in clinical practice in sleep centres
There is a well-established connection between mental health conditions, such as depression, and sleep disturbances. People with insomnia may have a tenfold higher risk of developing cMDE and among people with depression, 75% of sufferers have trouble falling or staying asleepi. Furthermore, misdiagnosis of major depressive disorder is estimated at upwards of 65% in the USii. Despite this, depression screening is not routinely included in sleep studies, even though up to 21% of people undergoing a sleep study have depressioniii.
Positive results from SAMDE study highlight MEB-001’s significant potential:
The objective of the study (ClinicalTrials.gov ID NCT05708222) was to use MEB-001 to detect the likelihood of a cMDE using Clinician Reporting Outcomes (CRO) assessment in individuals referred to a sleep clinic. A total of 400 patients were recruited across 15 sleep centres in the US. Out of these, 73 patients were excluded due to incomplete data or a split night/titration sleep study. MEB-001 also automatically identified 32 patients with significant anomalies in their sleep data. Notably, the MEB-001 algorithm was locked prior to the analysis ensuring no data from Phase 2 was used in its training.
Analysis of the results showed that MEB-001 reported promising performance across key parameters including sensitivity of 87% (95% CI 73-96%), specificity of 72% (95% CI 66-77%), positive predictive value (PPV) of 35% (95% CI 27-45%) and negative predictive value (NPV) of 97% (95% CI 93-99%) (refer table below).
These results provide considerable validation of MEB-001 and its ability in the screening and diagnosis of a current Major Depressive Episode (cMDE) in test subjects.
Management commentary:
Non-executive Chairman, David Trimboli said: “We are very excited to share the results from our Phase2 study, which highlight the effectiveness of MEB-001 in screening for cMDE. This is underscored by the significant increase in sensitivity coming from recent improvements to the underlying algorithm, which was increased from 71% in Phase 1, to 87% in Phase 2 – demonstrating that the algorithm can successfully identify more people with cMDE. Our very high NPV validates MEB-001’s potential as a screening test, with the likelihood of having depression when testing negative to be less than 3%.
“There is currently no screening undertaken for cMDE in sleep centres in the US or globally. Our most recent results reaffirm the potential of MEB-001 to become the first depression screening tool used in clinical practice in sleep centres and marks a significant achievement in the Company’s history.”
Click here for the full ASX Release
This article includes content from TrivarX, licensed for the purpose of publishing on Investing News Australia. This article does not constitute financial product advice. It is your responsibility to perform proper due diligence before acting upon any information provided here. Please refer to our full disclaimer here.
The Conversation (0)
Latest News
Interactive Chart
Latest Press Releases
Related News
TOP STOCKS
American Battery4.030.24
Aion Therapeutic0.10-0.01
Cybin Corp2.140.00