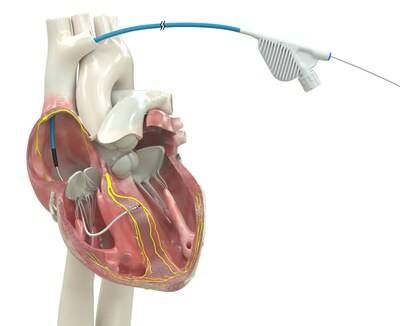
Built for high reliability, world's only lumenless defibrillation lead approved for placement in the left bundle branch area
Medtronic plc (NYSE: MDT), a global leader in healthcare technology, received U.S. Food and Drug Administration (FDA) approval for an expanded indication for the OmniaSecure™ defibrillation lead. Now approved for placement in the left bundle branch (LBB) area, the lead can be used for conduction system pacing (CSP), which closely mimics the heart's natural physiology. Additionally, patients in need of cardiac resynchronization may benefit from left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), a novel therapy that combines CSP with left-ventricular pacing to further improve patient outcomes.
The OmniaSecure defibrillation lead connects to an implantable cardioverter-defibrillator (ICD) or cardiac resynchronization therapy defibrillator (CRT-D) to treat potentially life-threatening ventricular tachyarrhythmias, ventricular fibrillation (VT/VF), and bradyarrhythmias. In addition to being the first defibrillation lead approved for placement in the LBB area, the OmniaSecure lead is the smallest diameter defibrillation lead on the market (4.7 French, or 1.66 mm) and the only one approved for adults and adolescent pediatric patients ages 12 and up.
Engineered based on the reliable Medtronic SelectSecure™ Model 3830 pacing lead − the first pacing lead to receive FDA approval for CSP in 2022 − the OmniaSecure defibrillation lead is built for high reliability and durability with its lumenless construction (no hollow channel on the inside), which enables the lead to be delivered via catheter for precise placement in the right ventricle, in addition to the LBB area. Conduction system pacing works by tapping into the heart's natural electrical system, providing patients needed therapy while avoiding cardiomyopathy or other complications sometimes associated with traditional pacing methods.1
"Conduction system pacing is a rapidly growing therapy for patients who need a pacemaker. Now, patients who require a defibrillator and pacing have an option that can safely deliver life-saving defibrillation therapy and activate the heart's natural electrical system to enable a more synchronous, physiologic pattern," said Trevor Cook, vice president and general manager of the Defibrillation Solutions business, which is part of the Cardiac Rhythm Management operating unit at Medtronic. "This approval underscores the versatility of the OmniaSecure defibrillation lead and supports its use across a variety of implant approaches to best serve a broad range of patients."
ICDs and CRT-Ds are the gold standard for preventing sudden cardiac arrest. For patients who require chronic pacing and defibrillation therapy, the OmniaSecure lead can offer treatment for dangerously fast arrhythmias while delivering conduction system pacing through the LBB area, when connected to a transvenous ICD or CRT-D, such as the Medtronic Cobalt™ and Crome™ family of devices. Patients indicated for CRT may benefit when OmniaSecure is paired with a left ventricular coronary sinus lead (i.e., biventricular or LOT-CRT configurations). For patients who do not require chronic pacing, the Medtronic extravascular Aurora EV-ICD™ system offers protection as the only ICD with a defibrillation lead positioned outside the vascular space that also provides anti-tachycardia pacing (ATP) in a single device.
FDA approval for the OmniaSecure defibrillation lead was supported by data from the global LEADR LBBAP trial (Lead Evaluation for Defibrillation and Reliability in Left Bundle Branch Area Pacing), which demonstrated the lead's safety and effectiveness. Late-breaking data from the study presented at Heart Rhythm 2025, and later published in Heart Rhythm, showed that the lead delivered high defibrillation success at implant (100%) and a low OmniaSecure-related major complication rate at three months (2.1%) when placed in the LBB area for physiologic pacing.2 Longer-term LBB area and LOT-CRT results from the study will be presented at HRS 2026.
"The robust body of clinical evidence from the LEADR LBBAP study supports the OmniaSecure defibrillation lead as a safe and effective way to activate physiologic pacing through the left bundle branch area or in applications like LOT-CRT, while also demonstrating reliable defibrillation success," said Pugazhendhi Vijayaraman, M.D., cardiac electrophysiologist at Geisinger Wyoming Valley Medical Center in Wilkes-Barre, Pa., and principal investigator of the LEADR LBBAP study.
The approval expands the comprehensive Medtronic portfolio of lumenless leads and accessories approved by FDA that enable conduction system pacing. This includes the SelectSecure Model 3830 pacing lead, now implanted in more than one million patients globally, as well as the recently cleared C320LBB delivery catheter and the 5944RL rotatable connector. Together, these CSP solutions deliver stability and long-term performance that further enhance procedural precision and versatility.
Following FDA approval for placement in traditional locations in the right ventricle, the OmniaSecure defibrillation lead launched commercially in the U.S. in January 2026.
About Medtronic
Bold thinking. Bolder actions. We are Medtronic. Medtronic plc, headquartered in Galway, Ireland, is the leading global healthcare technology company that boldly attacks the most challenging health problems facing humanity by searching out and finding solutions. Our Mission — to alleviate pain, restore health, and extend life — unites a global team of 95,000+ passionate people across more than 150 countries. Our technologies and therapies treat 70 health conditions and include cardiac devices, surgical robotics, insulin pumps, surgical tools, patient monitoring systems, and more. Powered by our diverse knowledge, insatiable curiosity, and desire to help all those who need it, we deliver innovative technologies that transform the lives of two people every second, every hour, every day. Expect more from us as we empower insight-driven care, experiences that put people first, and better outcomes for our world. In everything we do, we are engineering the extraordinary. For more information on Medtronic (NYSE: MDT), visit www.Medtronic.com and follow Medtronic on LinkedIn.
Any forward-looking statements are subject to risks and uncertainties such as those described in Medtronic's periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results.
Contacts:
Joey Lomicky
Public Relations
+1-763-526-2494
Ingrid Goldberg
Investor Relations
+1-763-505-2696
1 Padala SK and Ellenbogen KA. Card Electrophysiol Clin. 2021;13(4):755-84. |
2 Vijayaraman P, et al. Heart Rhythm. Published online October 17, 2025. |
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/medtronic-omniasecure-defibrillation-lead-is-the-first-lead-of-its-kind-to-receive-fda-approval-for-conduction-system-pacing-302720215.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/medtronic-omniasecure-defibrillation-lead-is-the-first-lead-of-its-kind-to-receive-fda-approval-for-conduction-system-pacing-302720215.html
SOURCE Medtronic plc