
November 10, 2025
InMed Pharmaceuticals (NASDAQ: INM) pairs innovative therapeutic development in Alzheimer’s, ophthalmology, and dermatology with recurring revenue from its BayMedica manufacturing division — giving investors rare small-cap biotech exposure to high-impact science with reduced financing risk.
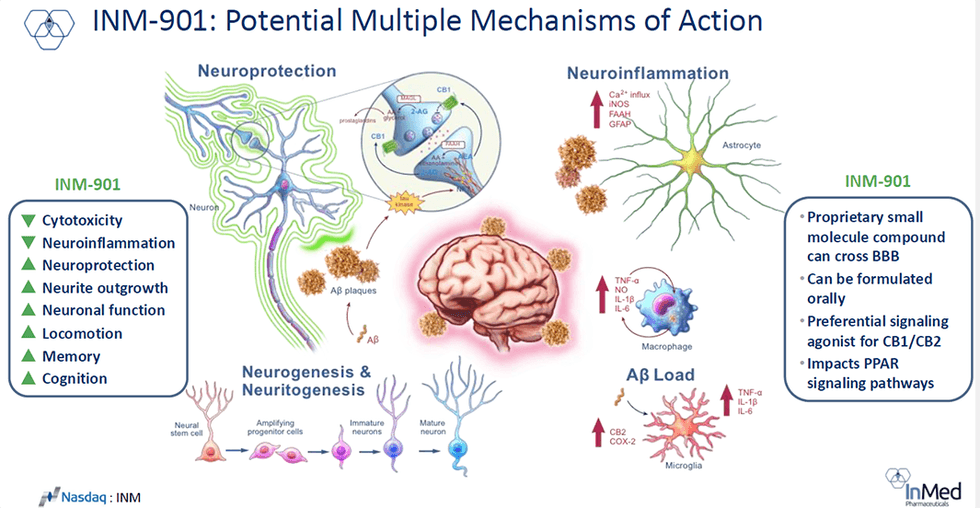
INM-901 takes a multi-pathway approach to Alzheimer’s, targeting several core drivers of the disease rather than just amyloid beta. In preclinical studies, it protected neurons, reduced inflammation, cleared toxic proteins, and improved cognition, aligning with the industry’s shift toward multi-target therapies.
InMed’s BayMedica subsidiary manufactures rare cannabinoids via chemical synthesis, rather than plant extraction, ensuring purity, consistency and scalability. The business generates approximately $5 million in annual revenue and ~40 percent gross margins, selling to the global health and wellness ingredient markets. This dual business model gives InMed a cash flow-supported R&D engine, enhancing sustainability and valuation resilience.
Investor Insight
InMed is a pharma innovator advancing proprietary small-molecule therapies in Alzheimer’s and ophthalmology, supported by a revenue-producing manufacturing arm. With cash exceeding its market cap and multiple near-term catalysts, it represents a compelling, undervalued biotech opportunity.
This InMed Pharmaceuticals profile is part of a paid investor education campaign.*
Click here to connect with InMed Pharmaceuticals (NASDAQ:INM) to receive an Investor Presentation
INM
INN Article Notification
The Conversation (0)
27 March
InMed Pharmaceuticals Receives Nasdaq Notification Regarding Minimum Bid Price Compliance
InMed Pharmaceuticals Inc. (NASDAQ: INM) ("InMed" or the "Company"), a pharmaceutical company developing a pipeline of disease-modifying small molecule drug candidates targeting CB1 and CB2 receptors, today announced the Company received a written notice from the Listing Qualifications... Keep Reading...
23 March
InMed Pharmaceuticals Announces Positive Data from Human Brain Organoid Neuroinflammation Models Supporting the INM-901 Alzheimer's Disease Program
Human Organoid Data Supports the Therapeutic Rationale for INM-901 Program Ahead of Human Clinical TrialsConsistent Anti-Inflammatory Effects Demonstrate Translation from Animal Models to Three-Dimensional Human Brain Tissue SystemsInMed Pharmaceuticals Inc. (NASDAQ: INM) ("InMed" or the... Keep Reading...
09 March
InMed Provides Update on Pharmaceutical Development Programs - Advancing Lead Drug Candidates Towards IND and Clinical Trial
Advancing Alzheimer's and Age-Related Macular Degeneration Programs Toward FDA Engagement and IND-Enabling ActivitiesTargeting Initiation of Phase 1 Clinical Trial in Alzheimer's Disease in 2027InMed Pharmaceuticals Inc. (NASDAQ: INM) ("InMed" or the "Company"), a pharmaceutical company focused... Keep Reading...
06 March
InMed Provides Update on BayMedica Operations and Strengthens Focus on Pharmaceutical Development Pipeline
InMed Pharmaceuticals Inc. (NASDAQ: INM) ("InMed" or the "Company"), a pharmaceutical company focused on developing a pipeline of disease-modifying small molecule drug candidates that target CB1CB2 receptors, today announced an update regarding BayMedica LLC ("BayMedica"), a wholly owned... Keep Reading...
11 February
InMed Reports Second Quarter Fiscal 2026 Financial Results and Provides Business Update
InMed Pharmaceuticals Inc. (NASDAQ: INM) ("InMed" or the "Company"), a pharmaceutical drug development company focused on developing a pipeline of proprietary small-molecule drug candidates for diseases with high unmet medical needs, today reports financial results for its second quarter of... Keep Reading...
Latest News
Interactive Chart
Latest Press Releases
Related News
TOP STOCKS
American Battery4.030.24
Aion Therapeutic0.10-0.01
Cybin Corp2.140.00
