Overview
A medical technology company based in Australia, Cleo Diagnostics (ASX:COV) is revolutionising women's healthcare with its disruptive cancer detection platform technology, through a simple blood test that can accurately detect ovarian cancer early – the leading cause of cancer-related deaths among women.
Approximately 50 percent of women will die within five years of an ovarian cancer diagnosis. The chances of survival beyond five years, however, increase with early detection. According to the American Cancer Society, only about 20 percent of ovarian cancers are diagnosed at an early stage, and more than 90 percent of women live beyond five years when the cancer is detected early.

With early diagnosis being key to a higher survival rate, ovarian cancer has become a target for biomarker research. And one particular biomarker holds promise.
Cleo’s technology is underpinned by the CXCL10 novel and patented biomarker, which was first identified as a small inflammatory molecule in ovarian cancer tissue sections. Subsequent research demonstrated that CXCL10 was overexpressed in ovarian cancers, but importantly not expressed in benign disease, and remains throughout the lifetime of the cancer. The biomarker effectively provides a robust indicator at all stages of cancer. Recognizing that early detection is a significantly unmet need in the clinical diagnostics market, Cleo Diagnostics is focused on bringing to market a simple blood test to accurately detect ovarian cancer early.
Cleo’s first clinical validation study for its ovarian cancer triage test has been published in the peer-reviewed international journal Cancers. The article concluded that Cleo’s ovarian cancer test was highly accurate with 95 percent sensitivity and 95 percent specificity, correctly discriminated malignant from benign samples, and has outperformed and was superior to current clinical methods. The second peer-reviewed dataset has also been published in the medical journal Diagnostics, which concluded that CLEO’s test has correctly identified most cancer cases that were missed by the standard marker CA125. It also eliminated the majority of “false positive” results caused by CA125 use, and it correctly identified the majority of patients with early-stage ovarian cancers.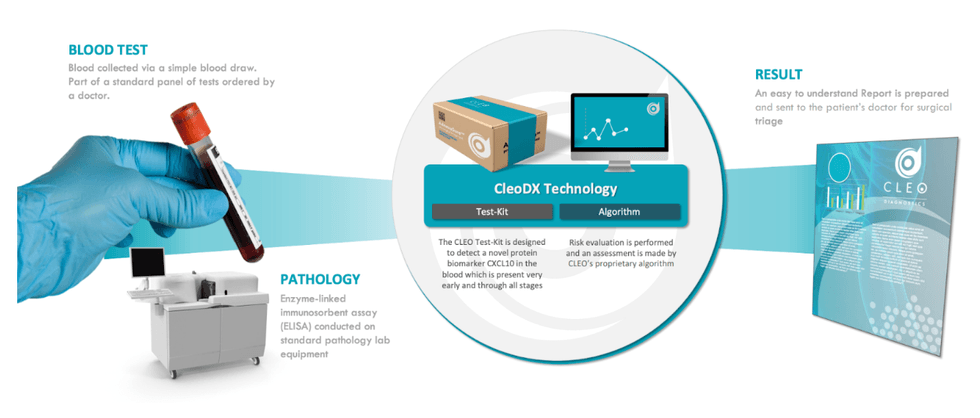
CLEO has appointed New York-based healthcare industry consultancy, HcFocus, to support the commencement of its US market access program. HcFocus will provide specialised and strategic expertise to assist CLEO in navigating the complexities of the US health system and regulatory environment.
The addressable market for a technology like this is compelling, and with a management team that brings to the table decades of leadership experience in the medical technology space, Cleo is well-positioned to leverage this market opportunity.
Cleo chief executive and executive director Dr. Richard Allman has over 30 years of experience in commercially focused scientific research and innovation. Throughout his career, Allman has overseen and expedited a product development pipeline covering no less than six major cancers, cardiovascular disease, type-2 diabetes and a commercially available COVID-19 test.
Chief scientific officer Dr. Andrew Stephens boasts an equally impressive resume. A career research scientist with two decades of experience in molecular and cellular biology, Stephens is named in over 60 academic publications and holds numerous patents in the cancer therapy and diagnostic space. Cleo’s blood test looks for a novel and patented biomarker in the blood called CXCL10, which was discovered by Stephens, the product of over ten years of scientific work at Monash Medical Centre's Hudson Institute of Medical Research.
There's also Professor Tom Jobling, Cleo's non-executive director and lead medical advisor. As the head of gynaecological oncology at Monash Health and visiting medical officer at the Peter MacCallum Cancer Centre, Jobling has been treating ovarian cancer for over thirty years. He was also the founding chairman of the Ovarian Cancer Research Foundation (OCRF)
Non-executive director Lucinda Nolan, meanwhile, brings significant business and strategic expertise to the table. Most recently, she served as the CEO of the Ovarian Cancer Research Foundation.
These experienced professionals, together with the other members of Cleo’s management and board, have developed a staged execution strategy focused on de-risking the pathway to the international screening market — ensuring that, although Cleo is still in its advanced R&D stage, its prospects for commercialisation remain incredibly promising.
Get access to more exclusive Medical Investing Stock profiles here

