Investor Insight
BriaCell Therapeutics is pioneering a new class of off-the-shelf personalized immunotherapies, exploring the potential to double overall survival in advanced breast cancer.
Company Highlights
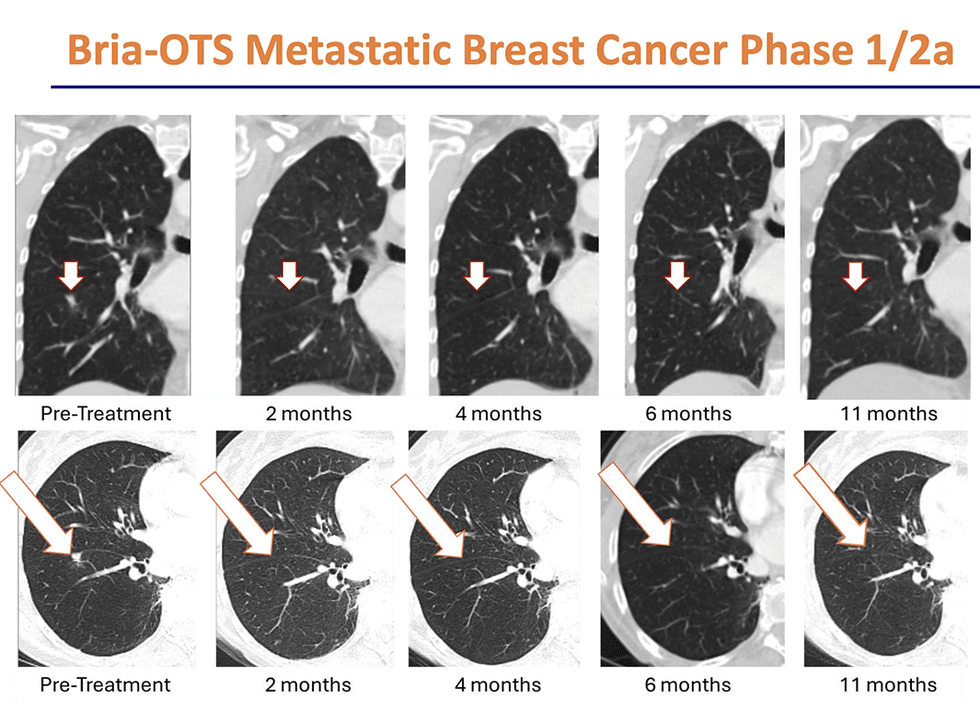
- Proven Survival Data: Clinical data have demonstrated an approximate doubling of overall survival compared to physician-placed chemotherapy.
- Personalized Immunotherapy: Its Bria-OTS™ platform uses a simple saliva test to provide HLA-matched therapy to over 99 percent of patients at a reasonable cost.
- Experienced Management: Leadership boasts a track record of 20 successful drug or device approvals, significantly de-risking the clinical and regulatory pathway.
- Expert Validation and Support: Recognized by Nature Medicine as a top transformative clinical trial of 2026 and supported by a grant from the National Cancer Institute.
Overview
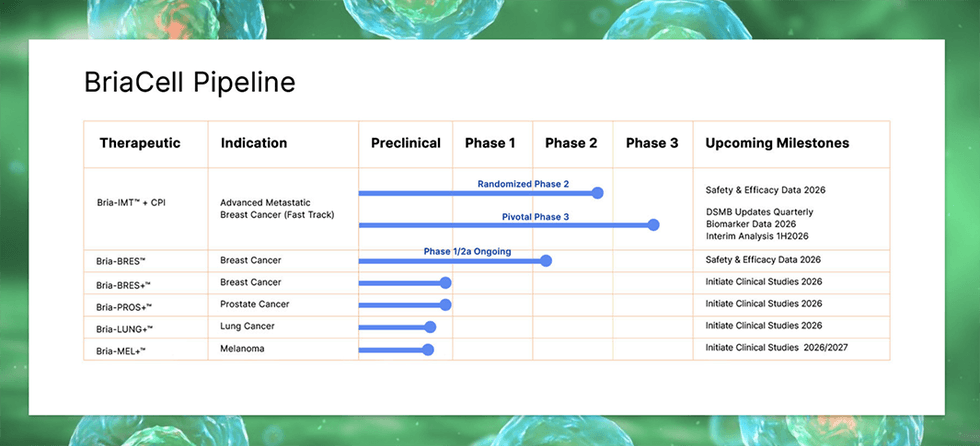
BriaCell Therapeutics (TSX:BCT,NASDAQ:BCTX) is a clinical-stage immuno-oncology company dedicated to transforming cancer care through the development of new classes of targeted immunotherapies. Its lead drug candidate is the Bria-IMT Cell Therapy, an off-the-shelf therapy designed to activate a patient’s immune system to target cancer cells without harming other cells. Bria-IMT™ is currently the subject of a pivotal Phase 3 study, under Fast Track designation, aimed at securing full approval from the US Food and Drug Administration (FDA).
The company is also advancing the Bria-OTS™ and Bria-OTS+™, a personalized off-the-shelf platform that aims to provide an innovative way to treat breast cancer and eventually expand to prostate and lung cancer and melanoma therapies.
BriaCell’s strategy is built on clinical validation and capital efficiency, with its seasoned team of scientists and clinicians having a combined record of 20 drug and device approvals.
*Disclaimer: This profile is sponsored by BriaCell Therapeutics ( TSX:BCT ). This profile provides information which was sourced by the Investing News Network (INN) and approved by BriaCell Therapeutics in order to help investors learn more about the company. BriaCell Therapeutics is a client of INN. The company's campaign fees pay for INN to create and update this profile.
INN does not provide investment advice and the information on this profile should not be considered a recommendation to buy or sell any security. INN does not endorse or recommend the business, products, services or securities of any company profiled.
The information contained here is for information purposes only and is not to be construed as an offer or solicitation for the sale or purchase of securities. Readers should conduct their own research for all information publicly available concerning the company. Prior to making any investment decision, it is recommended that readers consult directly with BriaCell Therapeutics and seek advice from a qualified investment advisor.