Overview
Beyond Medical Technologies (CSE:DOCT,FWB:7FM2)is a Canadian manufacturing company working to deliver essential safety equipment to protect citizens from airborne pathogens. The company’s flagship manufacturing facility is located in Delta, British Columbia, offers a fully-stable environment where it can produce technologically-enhanced personal protective equipment (PPE). Beyond Medical Technologies has invested significantly in the latest production and sterilization technology that it hopes will enable the production of high-grade protective medical equipment.
The COVID-19 crisis around the world has Beyond Medical Technologies focusing its manufacturing capacity on three core groups of users in need: hospitals, consumers and non-governmental organizations (NGOs). The company’s aim is to leverage technology to rapidly develop and produce masks and other protective equipment capable of protecting against common pathogens.
Mask use is recommended by public health authorities around the world as one of the leading means of preventing the spread of airborne viruses and diseases. During the COVID-19 pandemic, the widespread use of masks has been effective in reducing the “reproductive number” of the virus to below 1.0, a critical threshold.
“Societal norms and government policies supporting the wearing of masks by the public, as well as international travel controls, are independently associated with lower per-capita mortality from COVID-19,” concluded Virginia Commonwealth University’s Department of Ophthalmology in one study concerning mask use and global mortality rates.
Beyond Medical Technologies’ manufacturing facility in Delta, British Columbia is located close to major shipping hubs connected by rail, air and sea. The company anticipates this access to international distribution channels could allow it to quickly service regions around the world. Beyond Medical Technologies is targeting the global personal protective equipment (PPE) market, which is expected to grow to a total of US$84.7 billion by 2027 according to Grand View Research.
The company intends to continue to build out its manufacturing facility while developing proprietary products and increasing its existing portfolio through innovations and further acquisitions.
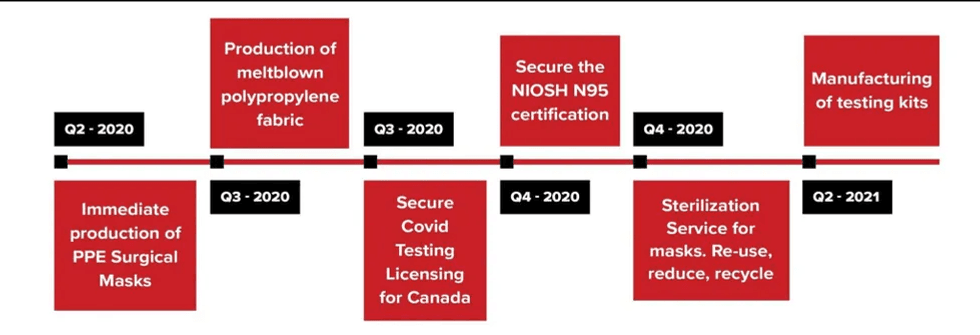
Beyond Medical Technologies prides itself on being a Canadian company with the goal of first protecting and satisfying the needs of Canadian citizens. Considering the severity of the COVID-19 pandemic, Beyond Medical Technologies is working to advance and scale its operations in order to support the global need for protective equipment as well.
The next catalyst for Beyond Medical Technologies is to secure a NIOSH N95 certification, while also exploring the potential of establishing a sterilization service for mask re-use.
Company Highlights
- State-of-the-art 5,078 square foot manufacturing facility is based in Delta, British Columbia with access to local shipping and distribution centers
- Targeting the global PPE market, expected to grow by US$84.7 billion by 2027 according to Grand View Research
- Working to produce medical-grade masks capable of preventing the spread of airborne pathogens
- Mask use has been proven by various studies to greatly reduce the reproductive number of the coronavirus
- Working towards vertical integration in the medical space by establishing melt-blown polypropylene fabric production
- The company’s wholly-owned subsidiary Micron Technologies achieves the Amazon Choice designation and begins to accept bitcoin payments for its Canada made face masks.
Get access to more exclusive Medical Device Investing Stock profiles here